Education & Training

Competencies
GS + trains in these areas of expertise:
- physiology of the cardiovascular and respiratory systems
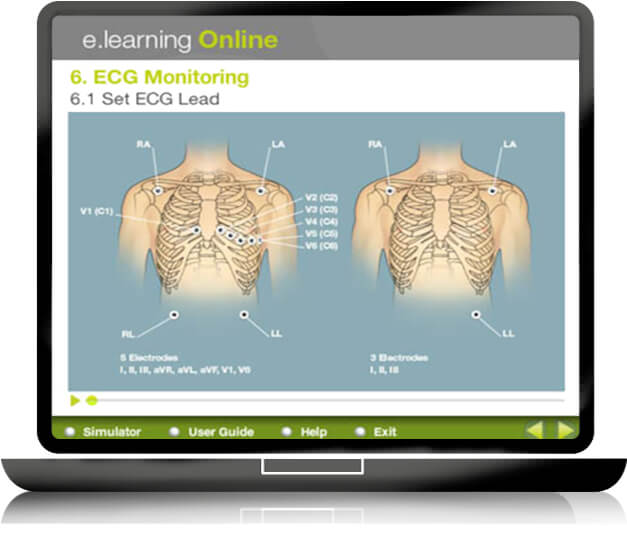
- ECG electrocardiogram and defibrillator
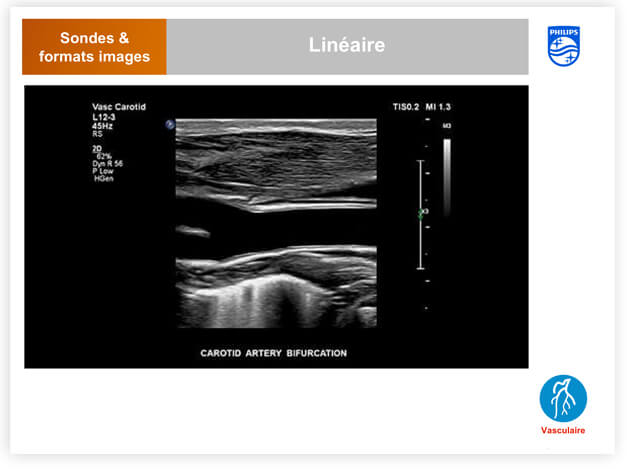
- ultrasound (technical, clinical, cardiac sonographer)
- physiology fetal and mother monitoring
- measurement parameters of monitoring
- patient safety and IEC 60601-1
- risk management (ISO 14971)
- maintenance preventive protocol
- electrical safety tests (IEC 62353)
- calibration and IEC 17025
- regulation (EU 2017/745)
- cyber security
- train the trainers


Training & lecture
GS + trains hospital biomedical technicians and the manufacturers’ field and application engineers.
We ensure the training courses for:
- the IFM Medical Training Institute of PHILIPS HEALTHCARE in Suresnes, France or directly on hospital sites
- Universities:
Cachan – Paris Sud (license)
Paul Sabatier – Toulouse (license & master)
Annecy (license)
ILIS Lille (master)

Evolution
The profession of biomedical technician a little known profession. This profession encompasses various services with the objective of patient safety. This profession is constantly evolving due to :
- technologies
- medical practices
- standards
- regulations (EU)
- IT
- softwares
- user interfaces
- human factors
Generally the lack of resources does not allow to make transformations and possible changes to make this job even more exciting.

Video & Photos
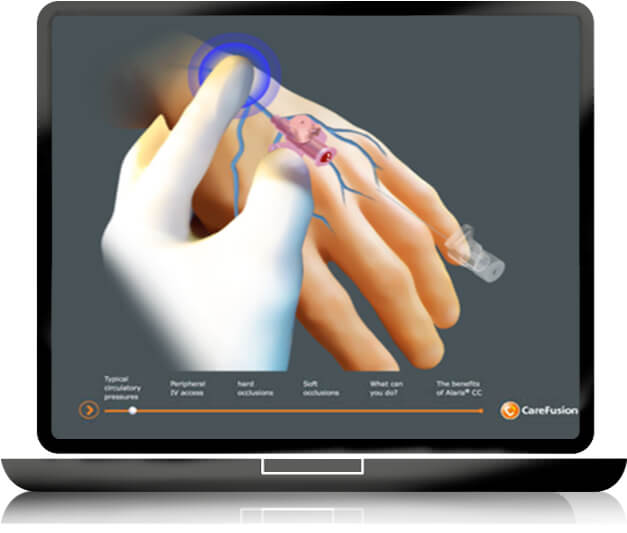
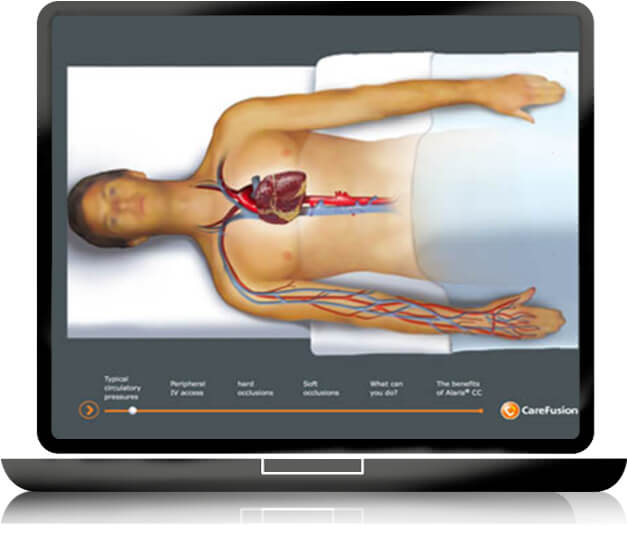
To gain time in the educational learning process of medical devices, GS + designs and produces short, easy to use and self-explanatory E LEARNING modules to meet the healthcare providers’ educational requirements:
GS + has the knowledge to produce and control the entire process:
- project management
- simplify the visuals
- define navigation ergonomics

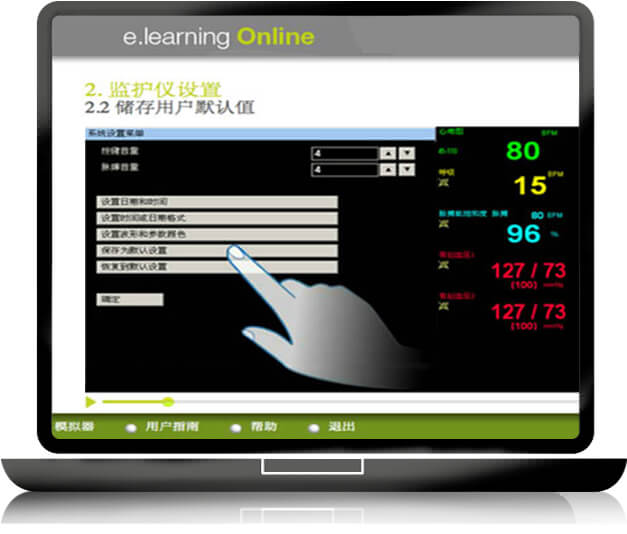
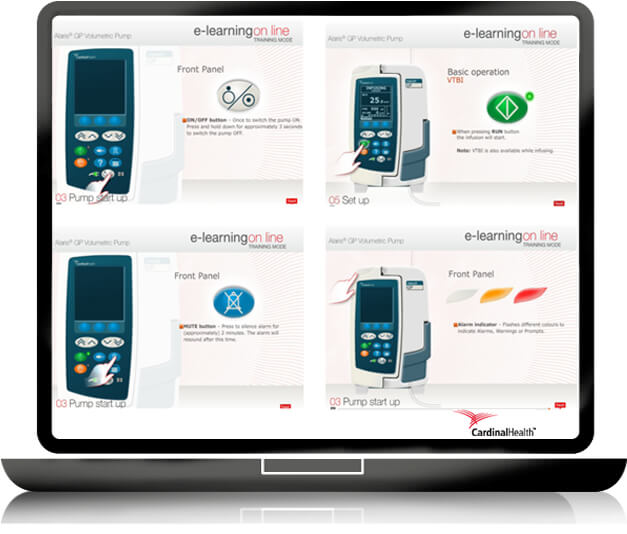
E learning
To gain time in the educational learning process of medical devices, GS + designs and produces short, easy to use and self-explanatory E LEARNING modules to meet the healthcare providers’ educational requirements:
GS + has the knowledge to produce and control the entire process:
- project management
- simplify the visuals
- define navigation ergonomics

User guide
Medical devices subject to “CE” marking oblige manufacturers to deliver together with the medical device:
- an instruction for use (IFU)
- a user manual
- a technical manual
GS + has the expertise to perfectly adapt the IFU to optimize the comprehension for the users.

Human factors
We provide global human factors, risk management, and design services to the MedTech and Pharma Industry.
We specialize :
- designing, testing, optimizing
- validating medical devices
- combination products
- digital health solutions to systematically create and optimize use-related safety
- usability
- user-acceptance in line with the current standards and guidelines

Translation
The commercialization of medical devices in Europe necessitates consolidating to a market with 14 different languages and cultures to which the user guides must adapt.
GS + responds to this problematic by simplifying the production of translations, reducing text and replacing it by diagrams, illustrations and pictograms, a process which makes the content and the educational advice perfectly understandable.

Assistante
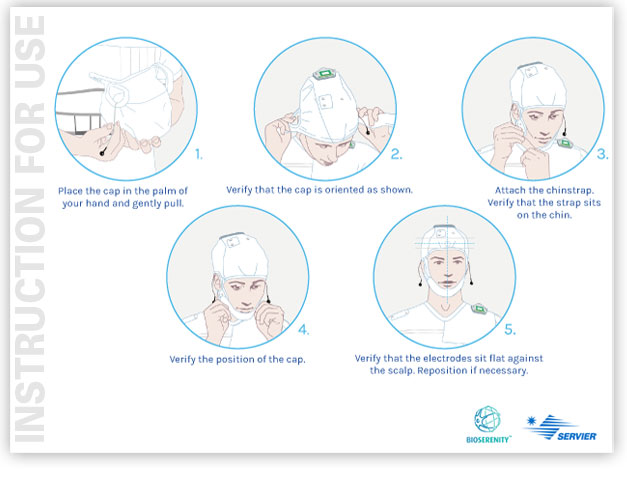
The PLUG & USE solution created and developed by GS +, assists and guides the users (the patient and the family) in the use of a medical device connected to HOME, to facilitate and ensure its start-up and to optimize its use.
Examples of connected devices:
- non invasive pressure
- ECG electrocardiogram
- SpO2 saturation
- fall detector
- electroencephalogram
- non invasive ventilation